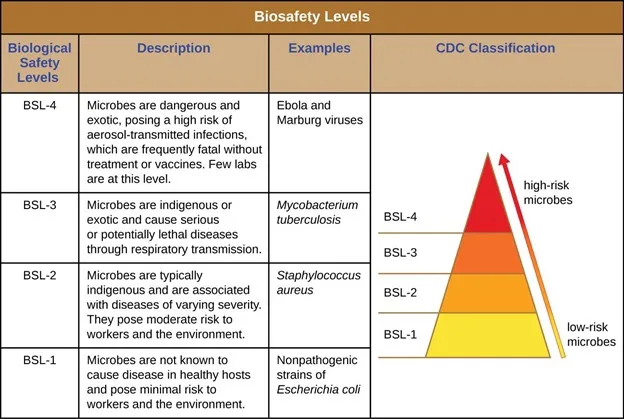
Due to their unique and potentially hazardous work, biosafety labs have special requirements in their construction, daily maintenance, and air pressure control. In fact, some labs must be kept in completely sterile conditions all the time. There are four BioSafety Levels (BSL): BSL 1 to 4, with 1 having the lowest risk of microbial spread and 4 having the highest risk (see table below).

In this blog post we will explore the unique aspects of autoclaving in BSL laboratories. First, we will take a look at some of the major differences between sterilization in hospitals versus in biosafety labs. Second, we will discuss bioshield frames, which surround autoclaves in many BSL 3 and 4 labs. And finally, we will explore the two major methods of disposing of autoclave waste, or effluent as it’s known, which are crucial in high-level labs to maintain safe conditions.
What is the Difference Between Hospital and Laboratory Contamination Concerns?
In a hospital’s Central Service Supply Department (CSSD)/Sterile Processing Department (SPD), the main concern is preventing the spread of infection through the medical devices that are processed. For example, it is the CSSD’s job to ensure complete sterility of surgical tool sets used in the operating room (OR), which means that all surgical tools must be completely cleaned, disinfected, and sterilized (autoclaved) before the next surgery. If not, the hospital runs the risk of exposing patients to harmful microorganisms and therefore, potentially causing a hospital-acquired infection (HAI).
Biosafety labs, by contrast, are afraid that the pathogens being studied will actually infect the outside surroundings through contamination. This is a crucial difference. For BSL labs, extra caution must be taken in order to prevent the spread of contaminants from room to room and to the outside environment. For example, the laboratory room is often kept at a specific set pressure in order to prevent the spread of contamination. And sometimes the room itself must be kept in constant sterile conditions when the risk of contamination is great.
What is a Bioshield Frame?
A bioshield frame is a biological seal that creates a complete hermetic seal around the autoclave, thus locking in all pathogens. In low level BSL labs, such as BSL 1-2, the goal of framing the autoclave is to prevent the passage of organisms like insects, and rodents, and other vermin, etc., whereas in the case of BSL 3 and 4 labs, the goal is to prevent the passage of microorganisms and/or gases. A bioshield system meets the bio-safety requirements for BSL3 and BSL4 labs. Exactly framing the hole in the wall where the autoclave is installed, the bioshield frame is made of a continuous, shock-absorbent neoprene seal and stainless-steel plates. In addition, the bioshield includes a jacket frame, which is composed of a fully welded metal flange and threaded studs. Any electrical connections to the autoclave pass through special sealed conduits that are part of the frame, thus maintaining completely sealed conditions.
By making sure that the autoclave is completely hermetically sealed into the wall, the BSL lab maintains full isolation of one side of the wall, where items enter the autoclave, which is totally separate from the other side of the autoclave, where these items emerge sterile. This pass-through autoclave system is absolutely crucial in BSL3 and BSL4 laboratories, which have much stricter sterility requirements due to the dangers of pathogen contamination.
For example, many biosafety labs have a “hot room,” which is the most protected and dangerous room of the lab since that is where scientists are working with the highest-risk microorganisms. This area must be protected and isolated from the rest of the lab by a pass-through autoclave, which is built into the wall and sealed by the bioshield frame. A hot room is up to 1000 times more clean, free of particles, and contamination than the operating room (OR) of the hospital!
HEPA Filter
High-Efficiency Particulate Air filters, known more commonly as HEPA filters, are a class of air filter designed to block 99.97% of particles that have the size of 0.3 µm (micrometers). These filters are used in the medical, scientific, and pharmaceutical industries where powerful filters are required to maintain sterile or near sterile conditions.
In BSL labs, HEPA filters are used to block the passage of microorganisms found in the autoclave effluent. The HEPA filter is the most common method of blocking pathogens inside the autoclave since the use of the HEPA filter can be validated by water intrusion tests and/or integrity tests. These are tests that confirm the proper performance of the filter and are run periodically.
Essentially the HEPA filter catches microorganisms. The filter is placed inside a special housing, which also functions as a steam sterilizer, and it is here that the trapped microorganisms are destroyed. The destruction of pathogens caught inside the filter not only eliminates them in order to maintain the sterile conditions of the surrounding lab, but it also allows for safe, sterile conditions of the next cycle inside the autoclave. In addition, this sterilization step allows for the safe disposal of the HEPA filter at the end of its lifetime.
Thermal Biohazard System
Less common than HEPA filters, the thermal biohazard system is another way to eliminate dangerous pathogens in the autoclave effluent. In this method, the effluent is mixed with high pressured steam, which enters via an ejector, and then together, the effluent and steam pass through a heat exchanger, which keeps the mixture at a high temperature for a sufficient time to ensure complete annihilation of all pathogenic matter. We can confirm the temperature of the outlet of this heat exchanger in order to verify that we have indeed sterilized the outlet before the effluent leaves the autoclave.
Other Special Considerations for BSL Labs
Sometimes biosafety labs, especially levels three and four, have special installation requirements. For example, autoclaving in these conditions may require clean piping and specialized components like pressure switches, safety valves, and two HEPA filters -- instead of the standard one filter -- in order to double confirm the successful trapping/sterilization of all microorganisms in the effluent.
In addition, some biosafety labs require the addition of a special tank, which is connected to the drainpipe of the autoclave. Instead of simply releasing all effluent liquid and gas to the general public sewage system, this tank is treated with chemicals that act as a further step along the way to ensure proper sterilization, which is also confirmed by chemical testing, before the effluent is finally released to the city drain.
BSL Take-Away Points
Biosafety labs must stop the spread of potential contamination of pathogens, which can be an immediate danger to people in the lab and the outside environment.
- The goal of biosafety sterilization is to prevent the spread of microorganisms to the outside environment whereas the goal of hospital (CSSD) sterilization is to prevent the spread of infection through maintaining sterile medical equipment used in surgery and other procedures.
- The bioshield frame creates a complete hermetic seal around the autoclave to maintain high levels of sterility around the autoclave and between the two differently qualified zones on either side of where the autoclave is mounted in the wall, for example both into and directly outside the “hot room.”
- All autoclave effluent, which is essentially the contaminated air extracted from the autoclave chamber before sterilization, must be blocked and then sterilized before leaving the autoclave by either the use of a HEPA filter or a thermal biohazard system.
- The HEPA filter blocks microorganisms in the filter, which are then sterilized inside the housing of the filter.
- The thermal biohazard system uses high-temperature, high-pressurized steam to sterilize the autoclave effluent.
- There may be more special considerations for autoclaving in BSL conditions, such as double HEPA filters or the addition of a special tank that uses chemicals as an extra safety measure before releasing the autoclave effluent into the main public sewage system
We hope you have learned from and enjoyed our discussion of the special considerations of autoclaving in biosafety laboratories. Please share your comments or questions below. We love hearing from our readers!