Air Filtration in Sterilization: HEPA Filters and Autoclave Air Handling
Air filtration in sterilization protects two things at once — the environment from biohazardous air leaving an autoclave or biocontainment lab, and the sterile load from contaminated air entering the chamber after sterilization. Like liquid filtration, it is a separation method: HEPA filters do not kill microorganisms, they retain them. The filter itself is then sterilized in place to render the captured organisms harmless.
This article explains HEPA filter performance, where air filtration sits inside the autoclave cycle, how it is used in BSL-3 and BSL-4 facilities, integrity testing, and applicable standards.
Why Air Filtration Matters in Sterilization
Two distinct safety problems drive the use of HEPA filters in steam sterilization and biocontainment:
- Air leaving the autoclave can be contaminated. During pre-vacuum air removal, the air pulled out of the chamber may contain viable bacteria, viruses, or biohazardous agents from the load. Releasing it untreated would expose operators and the surrounding environment.
- Air entering the chamber after sterilization can re-contaminate the load. During the air-admission phase at the end of the cycle, ambient air rushes in to break the vacuum. Without filtration, that air can deposit microorganisms onto the now-sterile load.
Air filtration in autoclaves designed for biohazard or BSL-3/BSL-4 work addresses both — a HEPA filter on the biohazard exhaust line, and a HEPA filter on the air inlet line.
How HEPA Filtration Works
HEPA (High-Efficiency Particulate Air) filters retain ≥99.97 % of airborne particles at 0.3 µm — the most penetrating particle size (MPPS). Particles smaller and larger than 0.3 µm are actually retained more efficiently because three different mechanisms operate:
| Mechanism | Effective for | How it works |
|---|---|---|
| Interception | Larger particles (~0.3–1 µm and above) | Particles touch a fiber as they flow past |
| Inertial impaction | Larger particles | Particles cannot follow the air stream around fibers and collide with them |
| Diffusion (Brownian motion) | Smallest particles (<0.1 µm) | Random motion increases collision probability |
The 0.3 µm MPPS is the worst case — too small for efficient interception/impaction and too large for efficient diffusion. Above 99.97 % retention at MPPS, HEPA filters typically retain >99.99 % of bacteria (≥0.5 µm), bacterial spores, and most viruses (which travel attached to larger droplet nuclei).
Higher classifications exist: ULPA (Ultra-Low Penetration Air) filters retain ≥99.999 % at 0.12 µm MPPS, used in cleanrooms and some BSL-4 applications.
Air Filtration in Autoclave Exhaust Systems
In an autoclave designed for biohazard sterilization, the cycle starts by evacuating air from the chamber to allow steam penetration. That exhaust air may carry viable pathogens.
A HEPA filter is installed on the biohazard outlet line. As the chamber is evacuated:
- All air passes through the HEPA filter before exiting the autoclave.
- Pathogens are retained on the upstream side of the filter media.
- Filtered, microorganism-free air is exhausted to the facility ventilation.
This is sometimes called "green sterilization" or environmental protection mode — it is not the kill step (the steam cycle that follows is), but it ensures no pathogens reach the lab environment during air removal.
Why Hydrophobic Membranes
HEPA filters used in autoclaves are constructed from hydrophobic (water-repelling) borosilicate glass-fiber or PTFE media. The hydrophobic surface:
- Does not pass liquid water.
- Resists wetting by chamber condensate.
- Avoids the blocking and bypass risks that a wetted filter would create.
To keep the filter dry and functional, the biohazard outlet is positioned in the upper-middle region of the chamber wall, away from where condensate pools at the chamber bottom.
Sterilizing the Filter
After the cycle, the HEPA filter holds whatever organisms it captured. The filter must itself be sterilized, which is accomplished one of two ways:
- Integrated cycle: the filter is enclosed in a steam-jacketed housing and is sterilized as part of every biohazard cycle.
- Dedicated filter-sterilization cycle: the housing functions as a small autoclave-within-an-autoclave, with its own steam supply and temperature sensor, run after the main cycle.
In high-containment labs, two HEPA filters in series may be used so that failure of one filter still leaves a second barrier in place.
Air Filtration on the Inlet Line — Protecting the Sterile Load
After sterilization and drying, the autoclave breaks vacuum by admitting air. If that air came directly from the lab, it would deposit airborne microorganisms onto the now-sterile load.
A second HEPA filter is installed on the air inlet line:
- All air entering the chamber post-sterilization passes through the inlet HEPA filter.
- The load remains sterile until the door is opened.
This is the equivalent of, and just as important as, the exhaust filter — but in the opposite direction. Both filters together are the standard configuration for biohazard-rated autoclaves.
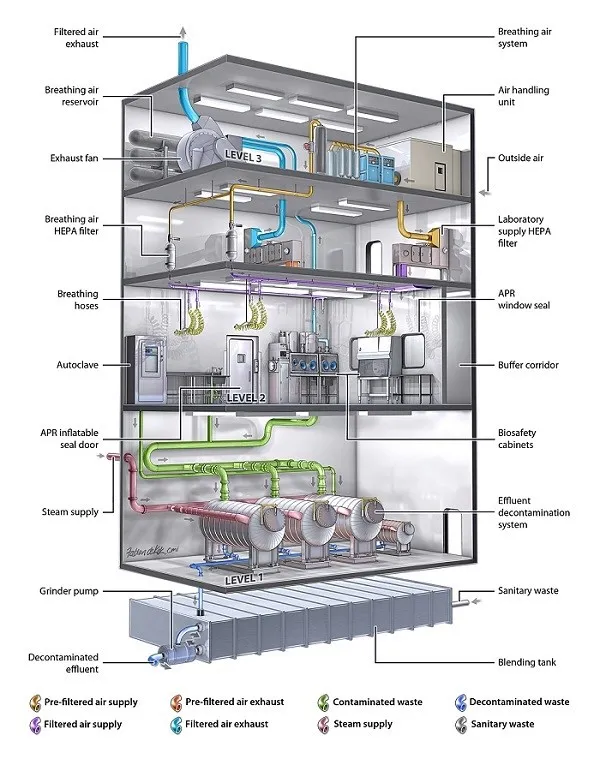
BSL-3 and BSL-4 Applications
Biocontainment laboratories (Biosafety Level 3 and 4) work with high-risk pathogens — Mycobacterium tuberculosis, hantaviruses, certain influenza strains (BSL-3); Ebola, Marburg, Lassa, smallpox (BSL-4). Their facility ventilation requires:
- HEPA filtration on all exhaust from the lab room and from any equipment that may release aerosols.
- Negative pressure in containment areas, with directional airflow toward the higher-containment zone.
- Double HEPA filters in series on BSL-4 exhaust for redundancy.
- Bag-in/bag-out (BIBO) filter housings that allow contaminated filters to be replaced without exposing maintenance personnel.
Pass-through autoclaves at the boundary of containment areas are dual-door units with HEPA-filtered air handling on both the contaminated and clean sides — a critical decontamination barrier between the lab and the rest of the building.
HEPA vs ULPA Filters
| Class | Retention | Particle size | Typical use |
|---|---|---|---|
| HEPA H13 | ≥99.95 % | 0.3 µm MPPS | Standard biohazard autoclave, BSL-2/BSL-3 |
| HEPA H14 | ≥99.995 % | 0.3 µm MPPS | BSL-3, ISO Class 5 cleanrooms |
| ULPA U15 | ≥99.9995 % | 0.12 µm MPPS | BSL-4, semiconductor cleanrooms |
| ULPA U16/U17 | ≥99.99995 % / ≥99.999995 % | 0.12 µm MPPS | High-end cleanroom, specialized containment |
For most autoclave applications, H13/H14 HEPA filters are sufficient. ULPA is used where the application requires retention of finer aerosols (semiconductor manufacturing) or where the absolute lowest particle penetration is mandated (some BSL-4 air handling).
Filter Integrity Testing
A HEPA filter that looks fine can still leak — at the gasket, at a tear in the medium, or because an installation error created a bypass. Integrity testing is required at installation and periodically thereafter.
| Test | Method | Use |
|---|---|---|
| Aerosol photometer (PAO/DEHS) scan | Challenge with a polydispersed oil aerosol upstream; scan downstream face for penetration | Standard installation and re-qualification test for HEPA in cleanrooms and biocontainment |
| DOP test (legacy) | Older variant using DOP aerosol | Largely replaced by PAO/DEHS due to DOP carcinogenicity concerns |
| Water Intrusion Test (WIT) | Challenge upstream side with water at controlled pressure; measure intrusion | Used for hydrophobic HEPA filters in autoclave applications, where the filter cannot be wetted in service |
| Pressure decay | Pressurize sealed housing and measure pressure drop over time | Quick installation check |
Integrity tests are required by ISO 14644-3 for cleanroom HEPA installations and by facility validation programs for BSL labs.
Maintenance and Replacement Schedules
HEPA filter life depends on dust loading and challenge. Typical service intervals:
- Visual / pressure-drop monitoring: continuous (delta-P transmitters)
- Integrity test: at installation, after any service, and at validated intervals (typically annually)
- Replacement: when pressure drop exceeds the design limit, when integrity fails, or per facility schedule (commonly every 3–5 years for clean exhaust applications, sooner for heavy bioburden duty)
In autoclaves, the HEPA housing is sterilized after every biohazard cycle, so the filter is not a contamination reservoir between cycles. Replacement is driven by clogging or integrity failure, not by accumulated bioburden.
Regulatory Standards
- ISO 14644-3 — Cleanrooms and associated controlled environments: Test methods, including HEPA installation and leak testing.
- EN 1822 / ISO 29463 — High-efficiency air filters (EPA, HEPA, and ULPA): Classification, performance testing, marking.
- ASHRAE 52.2 — Method of testing general ventilation air-cleaning devices.
- CDC/NIH BMBL (Biosafety in Microbiological and Biomedical Laboratories) — HEPA requirements for BSL-3 and BSL-4 facilities.
- WHO Laboratory Biosafety Manual — international biocontainment ventilation guidance.
- IEST RP-CC001 / RP-CC034 — recommended practice for HEPA/ULPA classification, leak testing.
FAQ
Is air filtration the same as sterilization?
Not exactly. HEPA filtration retains microorganisms; it does not kill them. In a biohazard autoclave, the HEPA filter on the exhaust line plus the steam cycle that sterilizes the filter together accomplish what is functionally a sterilization of the air leaving the chamber. In BSL labs, HEPA filters are part of the containment system, not a standalone sterilization method.
What is HEPA H13 vs H14?
H13 and H14 are filter classes under EN 1822 / ISO 29463. H13 retains ≥99.95 % of MPPS particles; H14 retains ≥99.995 %. H14 is required for higher-containment and cleaner cleanroom applications; H13 is sufficient for many standard biohazard autoclaves.
Why are autoclave HEPA filters hydrophobic?
The autoclave environment is wet — steam, condensate, and high humidity are all present. A hydrophilic filter would wet through and either block air flow or, worse, allow liquid to bypass the filter media at points of saturation. Hydrophobic borosilicate or PTFE media stay dry and retain their integrity through repeated cycles.
How are HEPA filters tested in autoclaves?
The standard for hydrophobic autoclave HEPA filters is the Water Intrusion Test (WIT) — water at a defined pressure is applied to the upstream side; intrusion is measured. WIT can be performed in situ with the filter still installed in the housing, which is a major advantage in containment installations.
What is the difference between HEPA and ULPA?
HEPA filters retain ≥99.97 % at 0.3 µm; ULPA filters retain ≥99.999 % at 0.12 µm. ULPA is used where the application requires retention of finer aerosols (semiconductor manufacturing) or where the absolute lowest particle penetration is mandated (some BSL-4 air handling). For most biohazard sterilization, HEPA is sufficient.
Do HEPA filters block viruses?
Yes — when carried on droplet nuclei or attached to other particles, viruses are retained at HEPA-class efficiency. Free, isolated virions in the 20–100 nm range are smaller than the 0.3 µm MPPS but are retained by diffusion at high efficiency. Bare-virion penetration through HEPA is far lower than the 0.03 % MPPS specification implies.
Conclusion
Air filtration in sterilization protects the environment during pre-vacuum air removal and protects the sterile load when air re-enters the chamber. The combination of hydrophobic HEPA filtration, integrated filter sterilization, and on-site integrity testing under ISO 14644-3 / EN 1822 makes biohazard autoclaves a reliable barrier for BSL-3 and BSL-4 work — and an essential layer alongside the steam sterilization cycle that performs the actual microbial kill. For liquid sterilization by membrane filtration see liquid filtration sterilization; for the broader method comparison see the sterilization methods overview.